脑血流监测
![]()
PSI 用于脑血流监测
高空间分辨率、更大可视区域和高帧率的独特组合使 PeriCam PSI 成为高质量脑部图像和监测快速变化的理想仪器。
脑血流量 (CBF) 的变化是许多神经系统疾病的特征,因此是神经科学领域众多研究的焦点。这些研究需要工具来研究这些变化,最好通过无创且实时的方法。PeriCam PSI 系统是一种基于激光散斑对比分析 (LASCA) 技术的血流灌注成像仪。LASCA 为研究微循环提供了前所未有的全新手段。它可以实现组织血流灌注的实时可视化,并将动态反应与空间分辨率相结合。它对灌注没有影响,因为不需要直接接触组织,也不需要显影剂或示踪元素。为了进一步提高其可用性,我们开发了专用的应用软件 PIMSoft。PSI 在多种小鼠模型中被广泛用于监测 CBF 和 CBF 的变化,以描述疾病病理和监测治疗效果。
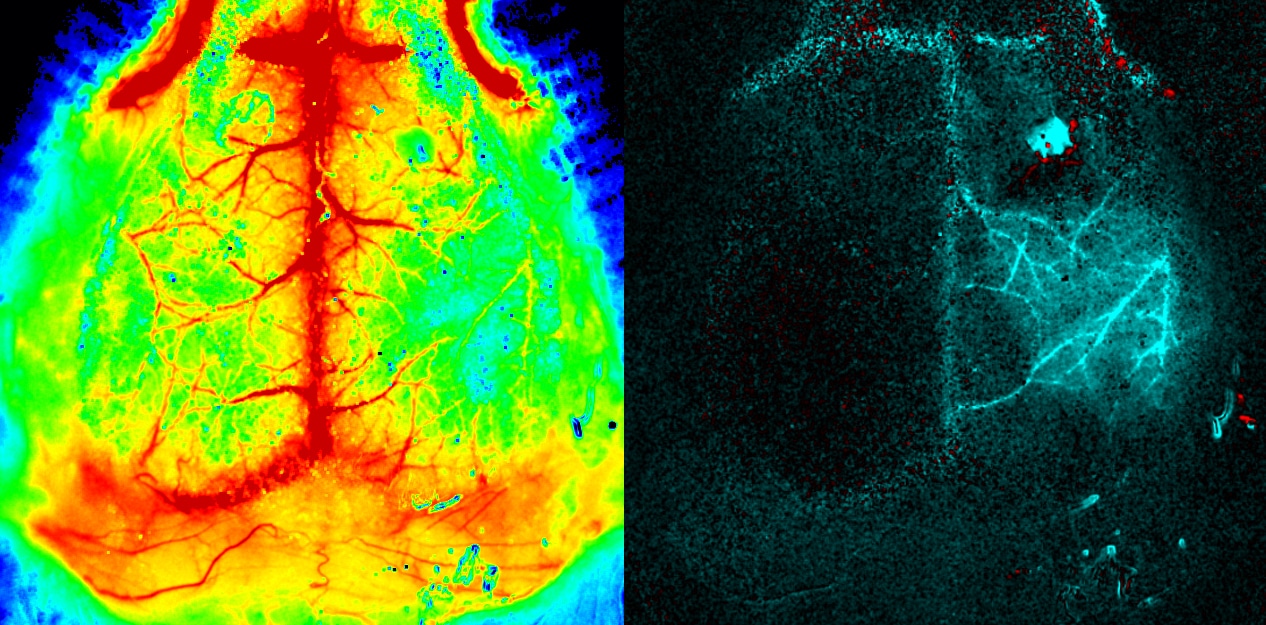
小鼠大脑。PeriCam PSI HR(高分辨率).由德国柏林夏里特医院的 Offenhauser 博士提供。
概述
PeriCam PSI 已被证明是脑部血流研究领域内一系列不同分领域的有
用工具。其中一个主要分领域是缺血性脑损伤的研究。可诱发缺血性和出血性卒中,使用 PeriCam PSI 确定卒中范围,并可跟踪卒中后随时间的动态过程。同样,慢性脑灌注不足和创伤性脑损伤模型也适合使用 PeriCam PSI 进行分析。
另一个分领域是脑部血流动力学变化的研究,高空间分辨率、更大可视区域和高帧率的结合使 PeriCam PSI 成为研究 CBF 快速变化的理想工具。该技术非常适合可视化皮质扩散性去极化中发生的灌注变化波,并且考虑到该应用,还开发了差分图像模式。最近,神经血管偶联也被证明是该技术的一个合理应用。
PSI 用于表征缺血性脑损伤
PSI 具有独特的功能,可以研究几种模型中的缺血性脑损伤,包括脑卒中、慢性脑灌注不足和创伤性脑损伤。
更大可视区域:整个大脑区域可视化,便于缺血性损伤的确认和表征
高空间分辨率:提供损伤的精确位置。插入感兴趣区域 (ROI) 可以测量损伤区域,并可用于跟踪损伤恢复情况。
恢复记录功能:通过在单个文件中捕获同一受试者的重复测量数据,简化了纵向研究的数据采集和分析,可以轻松比较相同 ROI 随时间发生的变化。
卒中
o 大脑中动脉闭塞 (MCAO) 模型 – 将细丝插入动脉以将血流封闭一段固定的时间(通常为 30-120 分钟),然后拔除细丝实现再灌注,即会导致缺血性脑卒中 1-9。
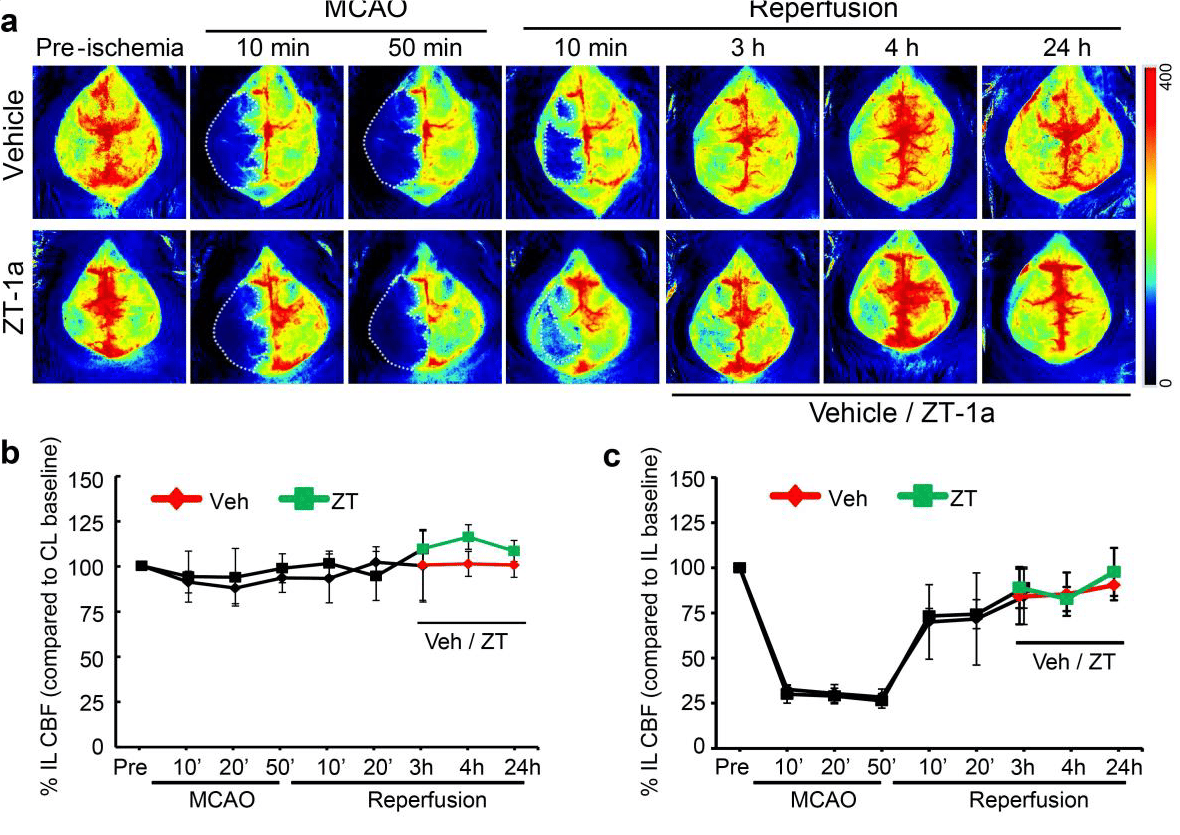
MCAO 前、MCAO 期间和 MCAO 后小鼠脑灌注图像以及处理和未处理动物同侧和对侧血流灌注的定量变化。
由匹兹堡大学神经科和匹兹堡神经退行性疾病研究所的 Dandan Sun 和 Iqbal H. Bhuiyan 提供。图片经《自然通讯》杂志许可转载,最初发表于 Zhang,J.,Bhuiyan,M.I.H.,Zhang,T. 等人的文章:Modulation of brain cation-Cl− cotransport via the SPAK kinase inhibitor ZT-1a.Nat Commun 11, 78 (2020). doi:10.1038/s41467-019-13851-6.
o 蛛网膜下腔出血 (SAH) 模型 – 插入细丝穿透大脑前动脉以导致卒中 10-13。
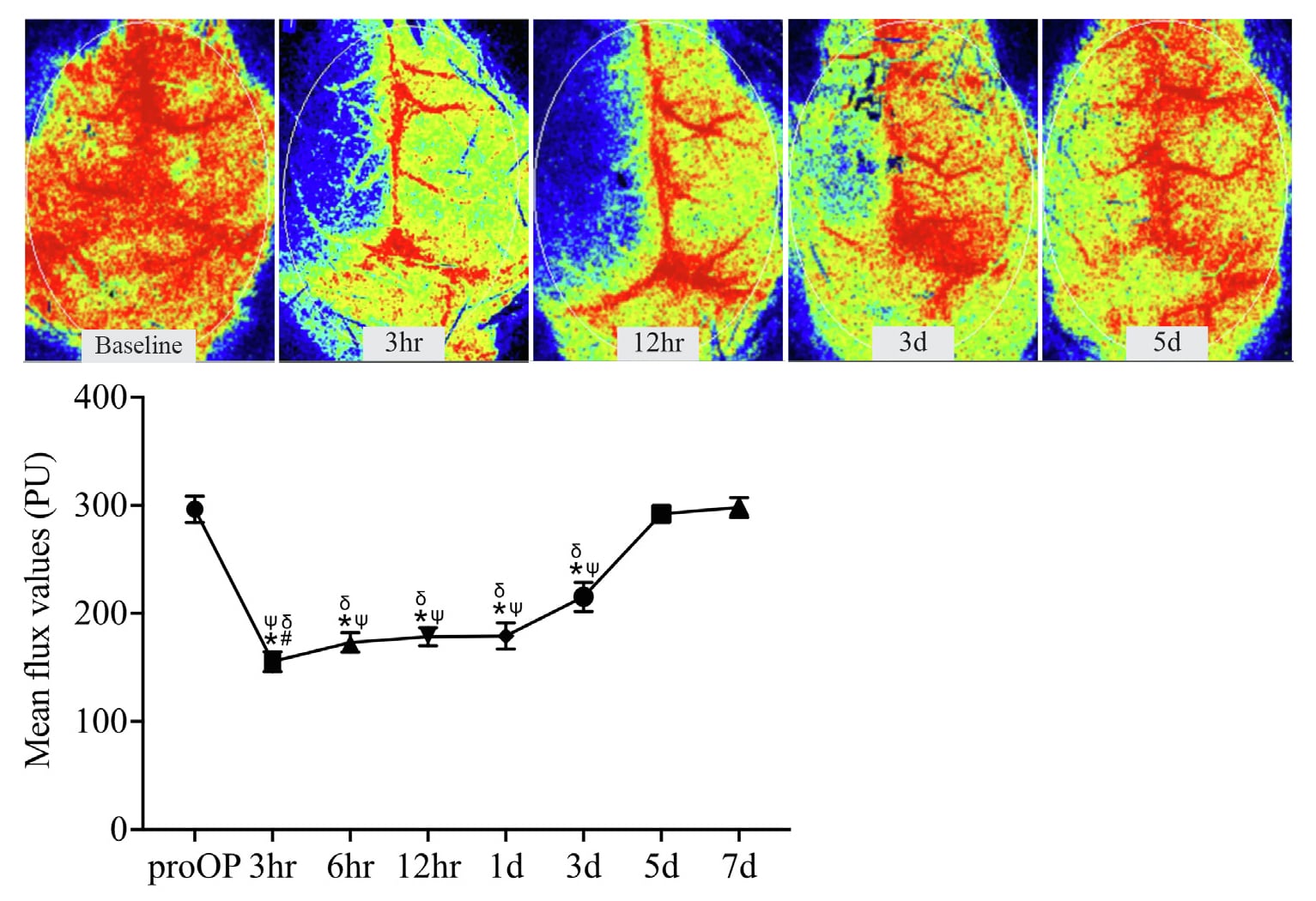
脑部研究 1727(2020)146566
SAH 损伤前后小鼠脑灌注图像。损伤后对同一只动物跟踪一周,以评估恢复情况。用 PSI HR 采集图像。
慢性脑灌注不足
o 双侧颈总动脉闭塞 (BCCAO) 模型 – 使用缝线或微弹簧圈双重结扎颈总动脉,以在双侧大脑半球创建缺血区域 14-20。
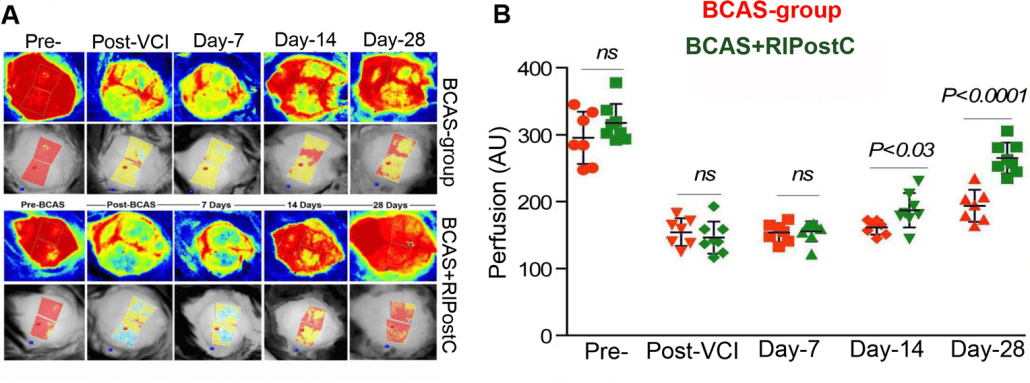
BCCAO 损伤前后小鼠脑图像的灌注图像以及处理和未处理动物血流灌注的定量变化。损伤后对动物进行一个月的跟踪。
由乔治亚摄政大学、乔治亚大学和 Charlie Norwood VA 医学中心的 Nasrul Hoda 博士提供。
图片经 Translational Stroke Research 杂志许可复制,最初发表于:Khan,M.B.,Hoda,M.N.,Vaibhav,K. 等人的文章:Remote Ischemic Postconditioning:Harnessing Endogenous Protection in a Murine Model of Vascular Cognitive Impairment.Transl.Stroke Res.6, 69–77 (2015). https://doi.org/10.1007/s12975-014-0374-6.
创伤性脑损伤
o 控制性脑皮质撞击 (CCI) 模型 – 用于对感觉运动皮质产生受控损伤的挫伤器械 21-23。
o 重物坠击/撞击加速模型 – 将重物坠击到无保护的颅骨上以造成脑损伤。
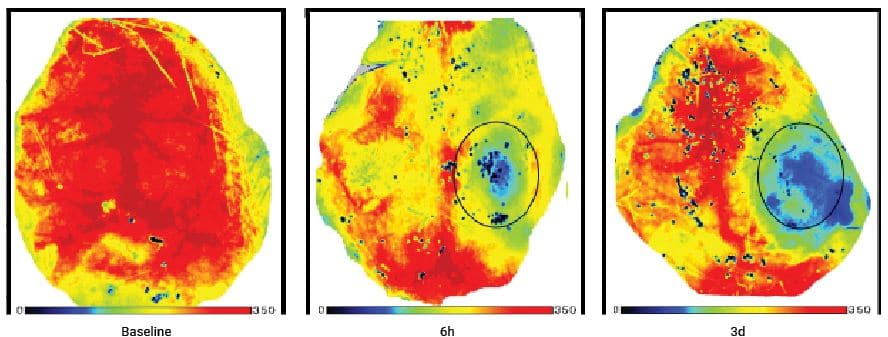
创伤性脑损伤前后小鼠脑部的灌注图像。损伤后对同一只动物跟踪 3 天。
由重庆医科大学附属第一医院神经外科 Han Liu 博士提供(中国重庆市,邮编 400016) H. Liu 等/《脑研究》(Brain Research) 杂志 1700(2018)118–125
PSI 用于表征脑部血流动力学变化
PSI 的独有功能使其能够用于研究刺激引起的 CBF 的微小动态变化,因为它能可视化灌注的空间和时间变化。异常反应是提示有认知功能障碍的早期体征。
高帧率和空间分辨率,使其可用于捕捉由于外部刺激导致的小结构中脑微血管系统的动态变化。
差分图像提供了更好的可视化效果,可以捕捉到 CBF 更细微的变化。
神经血管偶联 (NVC)
关键的稳态机制,确保充分调整 CBF 以增加激活神经元的氧和营养需求,产生功能性充血。NVC 在维持健康认知功能方面具有必不可少的作用。PSI 用于记录对坐骨神经 24 施加电刺激或胡须刺激 25-28 引起的躯体感觉皮层的血流动力学变化。NVC 受损导致年龄相关的认知功能下降。
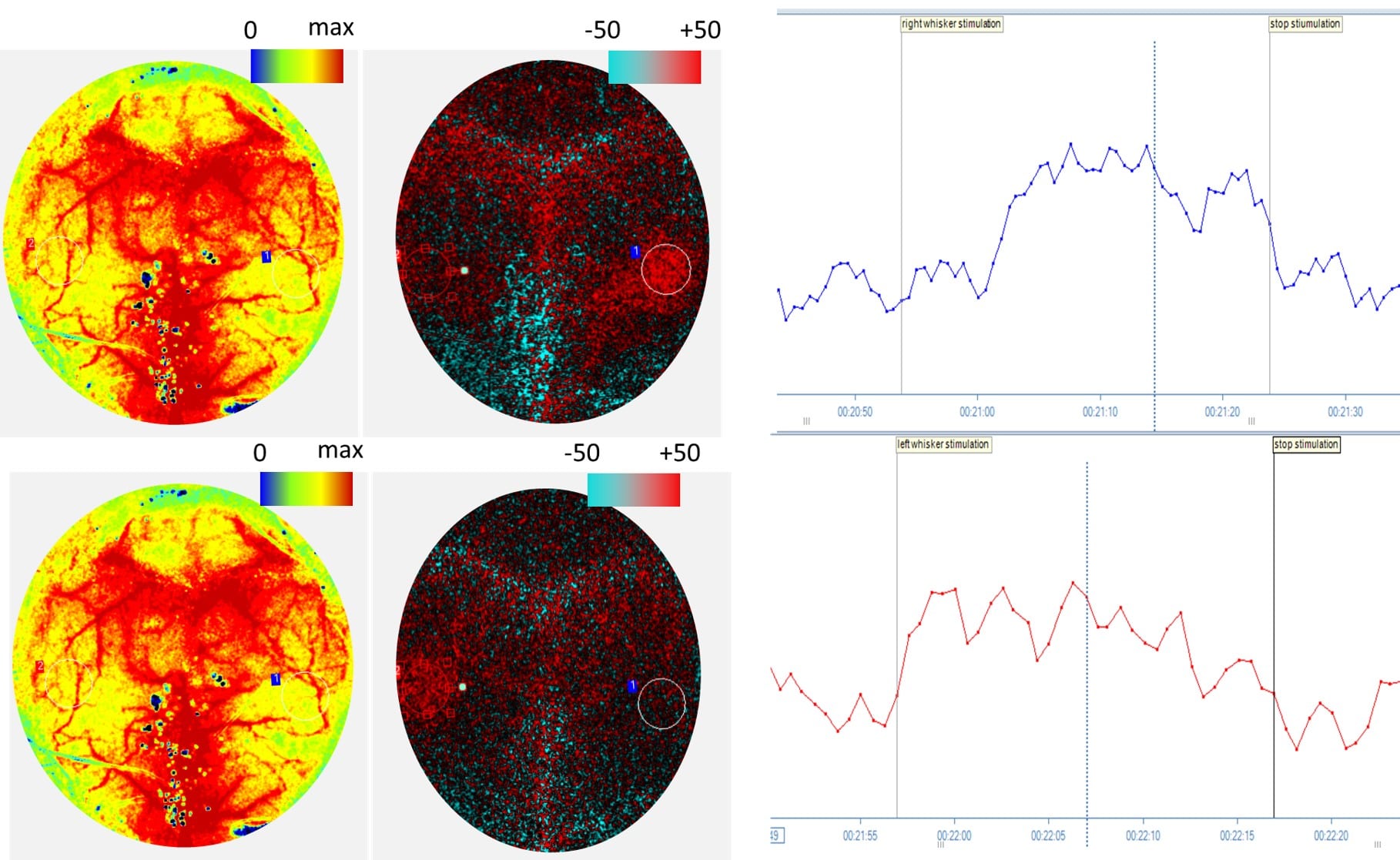
小鼠脑灌注图像显示为右侧(顶部)和左侧(底部)胡须 30 秒刺激期间的绝对和差分图像。刺激期间桶状皮层(预期会有灌注量增加)上 ROI 的 PIMSoft 图形数据。认知功能障碍导致由刺激引发的灌注增加下降。
由俄克拉荷马大学健康科学中心(俄克拉荷马州俄克拉荷马市)的 Stefano Tarantini 博士和 Zoltan Ungvari 博士提供。
皮层扩散性去极化 (CSD)
神经元跨膜离子梯度的突然、几乎完全破坏的波,会引起细胞毒性水肿,并在脑灰质中以约 3 mm/min 的速度传播,导致扩散性缺血。CSD 是急性脑损伤后神经退行性疾病的核心。PSI 用于记录由于针刺或局部施用高浓度钾溶液而引起的血液动力学变化 29-32。
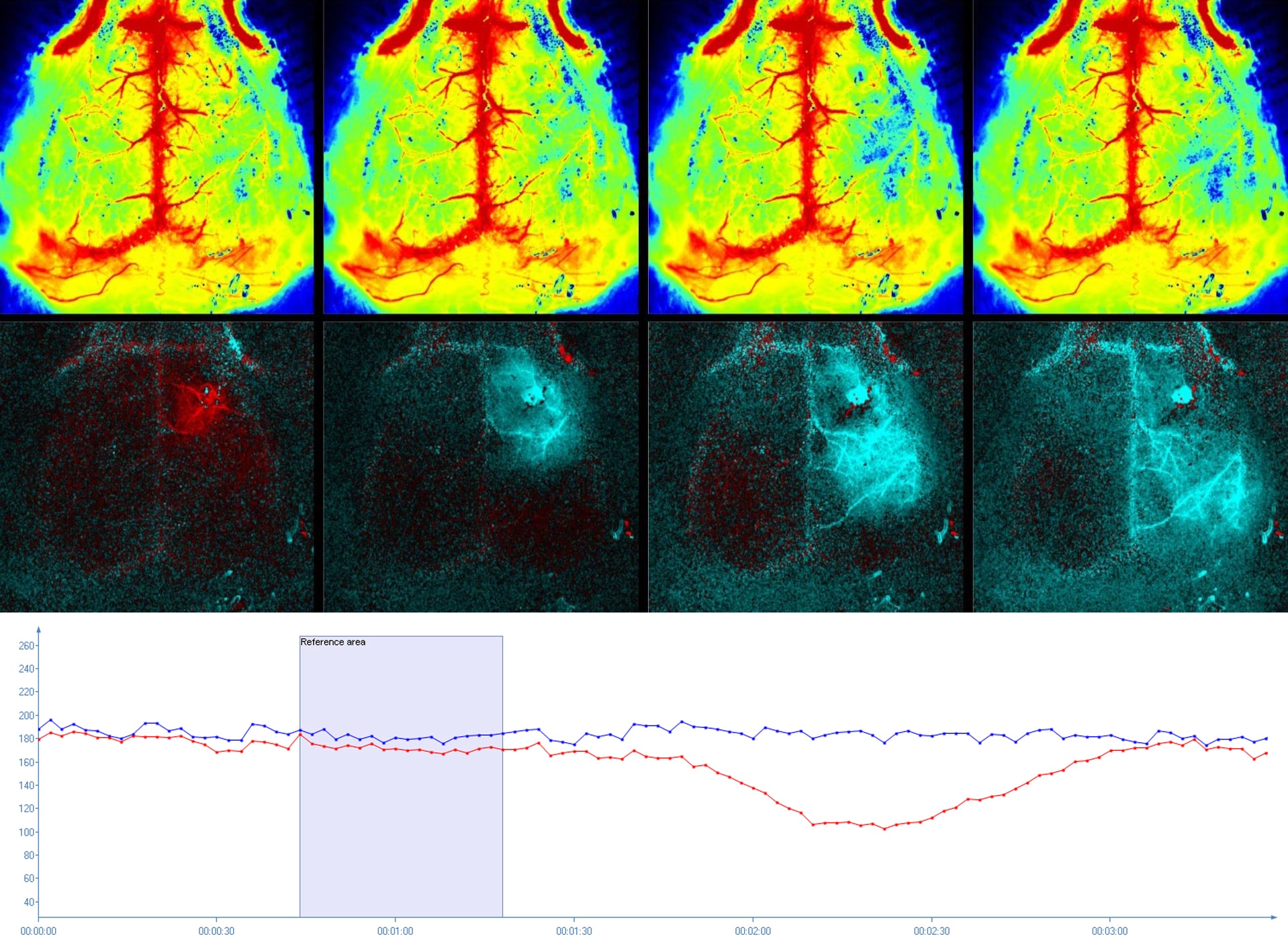
使用 PeriCam PSI HR 成像的小鼠大脑中的扩散性去极化。绝对灌注(顶行)可以可视化和量化(图),并且使用专门开发的差分图像模式(底行),可以一种令人惊叹的方式跟踪 SD 波引起的灌注变化。由德国柏林的夏尔特医院提供。
References
1. Morroniside promotes angiogenesis and further improves microvascular circulation after focal cerebral ischemia/reperfusion. T. Liu, B. Xiang, D. Guo, F. Sun, Re. Wei, G. Zhang, H. Aia, X.Tian, Z. Zhu, W. Zheng, Y. Wanga W.Wang. 2016, Brain Res Bull. , pp. 111-118.
2. C‐C Chemokine Receptor Type 5 (CCR5)‐Mediated Docking of Transferred Tregs Protects Against Early Blood‐Brain Barrier Disruption After Stroke. Peiying Li, Long Wang, Yuxi Zhou, Yu Gan, Wen Zhu, Yuguo Xia, Xiaoyan Jiang, Simon Watkins, Alberto Vazquez, Angus W. Thomson, Jun Chen, Weifeng Yu, Xiaoming Hu. 2017, Journal of the American Heart Association, p. e006387.
3. Endothelium-targeted overexpression of heat shock protein 27 ameliorates blood–brain barrier disruption after ischemic brain injury. Yejie Shi, Xiaoyan Jiang, Lili Zhang, Hongjian Pu, Xiaoming Hu, Wenting Zhang, Wei Cai, Yanqin Gao, Rehana K. Leak, Richard F. Keep, Michael V. L. Bennett, and Jun Chen. 2017, PNAS, Proceedings of the National Academy of Sciences, pp. E1243-E1252.
4. Brain ischemic preconditioning protects against ischemic injury and preserves the blood-brain barrier via oxidative signaling and Nrf2 activation. Tuo Yang, Yang Sun, Leilei Mao, Meijuan Zhang, Qianqian Li, Lili Zhang, Yejie Shi, Rehana K. Leak, Jun Chen, Feng Zhang. 2017, Redox Biology, pp. 323-337.
5. Brain-Derived Glia Maturation Factor β Participates in Lung Injury Induced by Acute Cerebral Ischemia by Increasing ROS in Endothelial Cells. Fei-Fei Xu, Zi-Bin Zhang, Yang-Yang Wang & Ting-Hua Wang. 2018, Neuroscience Bulletin, pp. 1077-1090.
6. The microRNA miR-7a-5p Ameliorates Ischemic Brain Damage by Repressing α-Synuclein. Kim T, Mehta SL, Morris-Blanco KC, Chokkalla AK, Chelluboina B, Lopez M, Sullivan R, Kim HT, Cook TD, Kim JY, Kim H, Kim C, Vemuganti R. 2018, Science Signaling, p. eaat4285.
7. Assessing the effects of Ang-(1-7) therapy following transient middle cerebral artery occlusion. M. M. C. Arroja, E. Reid, L. A. Roy, A. V. Vallatos, W. M. Holmes, S. A. Nicklin, L. M. Work & C. McCabe. 2019, Scientific Reports, p. 3154.
8. Modulation of brain cation-Cl− cotransport via the SPAK kinase inhibitor ZT-1a. Jinwei Zhang, Mohammad Iqbal H. Bhuiyan, Ting Zhang, Jason K. Karimy, Zhijuan Wu, Victoria M. Fiesler, Jingfang Zhang, Huachen Huang, Md Nabiul Hasan, Anna E. Skrzypiec, Mariusz Mucha, Daniel Duran, Wei Huang, Robert Pawlak, Lesley M. Foley, T. Kevin Hitc. 2020, Nature Communications.
9. Endothelium-targeted deletion of the miR-15a/16-1 cluster ameliorates blood-brain barrier dysfunction in ischemic stroke. Feifei Ma, Ping Sun, Xuejing Zhang, Milton H. Hamblin, and Ke-Jie Yin. 2020, Science Signaling.
10. Early Changes of Brain Perfusion After Subarachnoid Hemorrhage – the Effect of Sodium Nitroprusside. M Kolar, K Nohejlova, J Mares, J Pachl. 2016, Physiological Research, pp. S591-S599.
11. Changes of Cortical Perfusion in the Early Phase of Subarachnoid Bleeding in a Rat Model and the Role of Intracranial Hypertension. M Kolar, K Nohejlova, F Duska, J Mares, J Pachl. 2017, Physiological Research, pp. S545-S551.
12. Neuroprotective Effects of Nasopharyngeal Perfluorochemical Cooling in a Rat Model of Subarachnoid Hemorrhage. Mustafa Yavuz Samanci, Gennaro Calendo, Sandy T. Baker, Kadir Erkmen, Michael W. Weaver, Marla R. Wolfson. 2019, World Neurosurgery, pp. e481-e492.
13. Tauroursodeoxycholic acid prevents ER stress-induced apoptosis and improves cerebral and vascular function in mice subjected to subarachnoid hemorrhage. Xin Chen, Jianhao Wang, Xiangliang Gao, Ye Wu, Gang Gu, Mingming Shi, Yan Chai, Shuyuan Yue, Jianning Zhang. 2020, Brain Research, p. 146566.
14. TREM-2-p38 MAPK signaling regulates neuroinflammation during chronic cerebral hypoperfusion combined with diabetes mellitus. Jiawei Zhang, Yu Liu, Yaling Zheng, Yan Luo, Yu Du, Yao Zhao, Jian Guan, Xiaojie Zhang & Jianliang Fu. 2020, Journal of Neuroinflammation, Vol. 17.
15. Chronic Remote Ischemic Conditioning Is Cerebroprotective and Induces Vascular Remodeling in a VCID Model. Mohammad Badruzzaman Khan, Sherif Hafez, Md. Nasrul Hoda, Babak Baban, Jesse Wagner, Mohamed E. Awad, Hasith Sangabathula, Stephen Haigh, Mohammed Elsalanty, Jennifer L. Waller, David C. Hess. 1, 2018, Translational Stroke Research, Vol. 9, pp. 51-63.
16. Astroglial NF-kB contributes to white matter damage and cognitive impairment in a mouse model of vascular dementia. Raman Saggu, Toni Schumacher, Florian Gerich, Cordula Rakers, Khalid Tai, Andrea Delekate and Gabor C. Petzold. 4, 2016, Acta Neuropathologica Communications, Vol. 76.
17. Astrocyte-derived lipocalin-2 mediates hippocampal damage and cognitive deficits in experimental models of vascular dementia. Suk, Jae‐Hong Kim Pan‐Woo Ko Ho‐Won Lee Ji‐Young Jeong Maan‐Gee Lee Jong‐Heon Kim Won‐Ha Lee Ri Yu Won‐Jong Oh Kyoungho. 2017, Glia, pp. 1-20.
18. Cerebral microvascular dysfunction in metabolic syndrome is exacerbated by ischemia–reperfusion injury. Nathalie Obadia, Marcos Adriano Lessa, Anissa Daliry, Raquel Rangel Silvares, Fabiana Gomes, Eduardo Tibiriçá & Vanessa Estato. 67, 2017, BMC Neuroscience, Vol. 18.
19. The impact of early aerobic exercise on brain microvascular alterations induced by cerebral hypoperfusion. Marina Leardini-Tristão, Juliana Pereira Borges, Felipe Freitas, Raquel Rangel, Anissa Daliry,. 2017, Brain Research, Vol. 1657, pp. 43-51.
20. The Role of Autophagy in the Correlation Between Neuron Damage and Cognitive Impairment in Rat Chronic Cerebral Hypoperfusion. Wenying Zou, Yufei Song, Yumei Li, Yu Du, Xiaojie Zhang, Jianliang Fu. 1, 2018, Molecular Neurobiology, Vol. 55, pp. 776-791.
21. Clinical and Basic Evaluation of the Prognostic Value of Uric Acid in Traumatic Brain Injury. Liu H, He J, Zhong J, Zhang H, Zhang Z, Liu L, Huang Z, Wu Y, Jiang L, Guo Z, Xu R, Chai W, Huo G, Sun X, Cheng C. 10, 2018, International Journal of Medical Sciences, Vol. 15, pp. 1072-1082.
22. Selective activation of cannabinoid receptor-2 reduces neuroinflammation after traumatic brain injury via alternative macrophage polarization. Molly Braun, Zenab T.Khan, Mohammad B.Khan, Manish Kumar, Ayobami Ward, Bhagelu R.Achyut, Ali S.Arbab, David C.Hess, Md. Nasrul Hoda, Babak Baban, Krishnan M.Dhandapani, Kumar Vaibhav. 2018, Brain, Behavior, and Immunity, Vol. 68, pp. 224-237.
23. Evolution of cerebral perfusion in the peri-contusional cortex in mice revealed by in vivo laser speckle imaging after traumatic brain injury. Han Liu, Junchi He, Zhaosi Zhang, Liu Liu, Gang Huo, Xiaochuan Sun, Chongjie Cheng. 2018, Brain Research, Vol. 1700, pp. 118-125.
24. Regulation of cortical blood flow responses by the nucleus basalis of Meynert during nociceptive processing. Thierry Paquette, Ryota Tokunaga, Sara Touj, Hugues Leblond, Mathieu Piché. 149, 2019, Neuroscience Research, pp. 22-28.
25. Demonstration of impaired neurovascular coupling responses in TG2576 mouse model of Alzheimer’s disease using functional laser speckle contrast imaging. Stefano Tarantini, Gabor A. Fulop, Tamas Kiss, Eszter Farkas, Dániel Zölei-Szénási, Veronica Galvan, Peter Toth, Anna Csiszar, Zoltan Ungvari, Andriy Yabluchanskiy. 4, 2017, GeroScience, Vol. 39, pp. 465-473.
26. Overexpression of catalase targeted to mitochondria improves neurovascular coupling responses in aged mice. Anna Csiszar, Andriy Yabluchanskiy, Anna Ungvari, Zoltan Ungvari & Stefano Tarantini. 5, 2019, GeroScience, Vol. 41, pp. 609-617.
27. Pharmacological or genetic depletion of senescent astrocytes prevents whole brain irradiation–induced impairment of neurovascular coupling responses protecting cognitive function in mice. Andriy Yabluchanskiy, Stefano Tarantini, Priya Balasubramanian, Tamas Kiss, Tamas Csipo, Gábor A. Fülöp, Agnes Lipecz, Chetan Ahire, Jordan DelFavero, Adam Nyul-Toth, William E. Sonntag, Michal L. Schwartzman, Judith Campisi, Anna Csiszar & Zoltan Ungvari. 2020, GeroScience.
28. Treatment with the mitochondrial-targeted antioxidant peptide SS-31 rescues neurovascular coupling responses and cerebrovascular endothelial function and improves cognition in aged mice. Stefano Tarantini, Noa M. Valcarcel-Ares, Andriy Yabluchanskiy, Gabor A. Fulop, Peter Hertelendy, Tripti Gautam, Eszter Farkas, Aleksandra Perz, Peter S. Rabinovitch, William E. Sonntag, Anna Csiszar, Zoltan Ungvari. 2, 2018, Aging Cell, Vol. 17, p. e12731.
29. The antagonism of prostaglandin FP receptors inhibits the evolution of spreading depolarization in an experimental model of global forebrain ischemia. Dániel P. Varga, Írisz Szabó, Viktória É. Varga, Ákos Menhyárt, Orsolya M. Tóth, Mihály Kozma, Armand R. Bálint, István A. Krizbai, Ferenc Bari, Eszter Farkas. 2020, Neurobiology of Disease, Vol. 137, p. 104780.
30. Na+/K+-ATPase α isoform deficiency results in distinct spreading depolarization phenotypes. Clemens Reiffurth, Mesbah Alam, Mahdi Zahedi-Khorasani, Sebastian Major and Jens P Dreier. 3, 2019, Journal of Cerebral Blood Flow & Metabolism, Vol. 40, pp. 622-638.
31. Systemic administration of l-kynurenine sulfate induces cerebral hypoperfusion transients in adult C57Bl/6 mice. Dániel Péter Varga, Ákos Menyhárt, Tamás Puskás, Ferenc Bari, Eszter Farkas, Zsolt Kis, László Vécsei, József Toldi, Levente Gellért. 2017, Microvascular Research, Vol. 114, pp. 19-25.
32. Ansari MZ, Kang EJ, Manole MD, Dreier JP, Humeau-Heurtier A. Monitoring microvascular perfusion variations with laser speckle contrast imaging using a view-based temporal template method. s.l. : Microvascular Research, 2017. pp. 49-59. Vol. 111.
请填写表单,以获取更多信息
免责声明:Perimed 网站上的产品并非在全球所有市场均获准销售。



